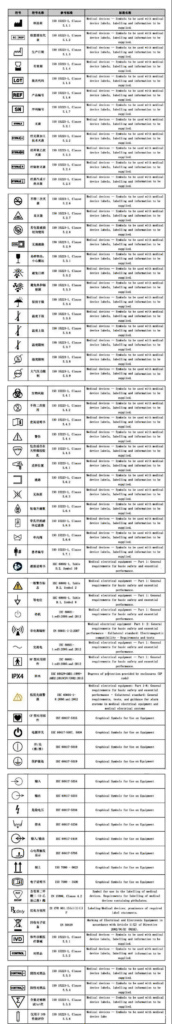
Medical device labels refer to the text, graphics, and symbols affixed to medical devices or their packaging that identify product features and indicate safety warnings. Medical device labels are an important vehicle for displaying information such as product safety, effectiveness, and key technical features. Currently, many medical device manufacturers, distributors, and users prioritize device quality control while neglecting the regulations governing medical device labeling.
Failure to properly label may pose a safety hazard to medical device users, and device manufacturers and operators may also be punished.
Regulatory requirements
Article 88 of the Regulations on the Supervision and Administration of Medical Devices: (2) Production and operation of medical devices whose instructions and labels do not comply with the provisions of these Regulations; (3) Failure to transport or store medical devices in accordance with the instructions and labeling requirements of medical devices. The department responsible for drug supervision and administration shall order rectification and impose a fine of not less than RMB 10,000 and not more than RMB 50,000; if rectification is refused, a fine of not less than RMB 50,000 and not more than RMB 100,000 shall be imposed; in serious cases, the production and operation shall be suspended, and the original issuing department shall revoke the medical device production license and medical device business license. The legal representative, principal person in charge, directly responsible supervisor and other responsible personnel of the illegal unit shall have their income from the unit during the period of the illegal act confiscated, and a fine of not less than 30% and not more than 2 times the income obtained shall be imposed, and they shall be prohibited from engaging in medical device production and operation activities for 5 years.
Regulatory requirements
1. Product name, model, and specifications;
2. Medical device registration certificate number or filing certificate number;
3. The name, address, and contact information of the registrant or record-filer. For imported medical devices, the name, address, and contact information of the agent shall also be stated;
4. The name, domicile, production address, contact information, and production license number or production registration certificate number of the manufacturer. In the case of entrusted production, the name, domicile, production address, production license number or production registration certificate number of the entrusted enterprise shall also be indicated;
5. Production date, expiration date or use-by date;
6. Power connection conditions and input power;
7. Necessary warnings and precautions;
8. Special storage or handling conditions or instructions;
9. Graphics, symbols and other relevant information that should be marked according to product characteristics;
10. Medical devices that may damage or negatively impact the environment during use should have warning signs or Chinese warning instructions on their labels;
11. The labels of medical devices that emit radiation or emit radiation shall include warning signs or Chinese warning instructions.
Note: If the medical device label cannot indicate all of the above information due to limited space or size, at least the product name, model, specifications, production date and service life or expiration date should be indicated, and the label should clearly state "See the instructions for other details."
Main regulatory requirements for medical devices
1. "Medical Device Supervision and Administration Regulations" Order No. 739 of the State Council of the People's Republic of China (effective from June 1, 2021): Article 39: The instructions and labels of medical devices shall indicate the following: (1) Generic name, model, and specifications; (2) Name, address, and contact information of the medical device registrant, record holder, and entrusted manufacturer; (3) Production date, service life, or expiration date; (4) Product performance, main structure, and scope of application; (5) Contraindications, precautions, and other matters requiring warnings or reminders; (6) Installation and use instructions or diagrams; (7) Maintenance and care methods, special transportation and storage conditions and methods; (8) Other matters that must be indicated as required by the product technical requirements. Class II and Class III medical devices shall also be marked with the medical device registration certificate number. Medical devices used by individual consumers shall also have special instructions for safe use. Article 57: Imported medical devices shall be medical devices that have been registered or recorded in accordance with the provisions of Chapter II of these Regulations. Imported medical devices shall have instructions and labels in Chinese. Instructions and labels must comply with the provisions of these Regulations and relevant mandatory standards. The instructions must clearly indicate the place of origin of the medical device and the name, address, and contact information of the domestic legal entity designated by the overseas medical device registrant or recorder. Medical devices without Chinese instructions or labels, or with instructions or labels that do not comply with the provisions of this article, shall not be imported.
2. "Regulations on the Administration of Instructions and Labels for Medical Devices" Order No. 6 of the State Food and Drug Administration (effective from October 1, 2014) Article 2: All medical devices sold and used within the territory of the People's Republic of China shall be accompanied by instructions and labels in accordance with the requirements of these regulations. Article 4: The contents of medical device instructions and labels shall be consistent with the relevant content of the registration or filing. The content of medical device labels shall be consistent with the relevant content of the instructions. Article 9: The text content of medical device instructions and labels shall be in Chinese, and the use of Chinese shall comply with the national general language and writing standards. Medical device instructions and labels may be supplemented with other languages, but the Chinese expression shall prevail.
What should not be included in the tag
(1) Containing assertions or guarantees of efficacy such as "best efficacy", "guaranteed cure", "guaranteed cure", "radical cure", "immediate effect", "completely non-toxic and side effects";
(2) Containing absolute language and expressions such as "highest technology", "most scientific", "most advanced", "best";
(3) Describing cure rate or efficiency;
(4) Comparing the efficacy and safety of other companies' products;
(5) Containing promise language such as "insurance company guarantee" or "invalid refund";
(6) Using the name or image of any organization or individual to prove or recommend;
(7) Containing misleading statements that make people feel that they have a certain disease, or misleading statements that make people misunderstand that not using the medical device will cause a certain disease or aggravate the disease, as well as other false, exaggerated and misleading content;
(8) Other content prohibited by laws and regulations;
(9) Descriptions unrelated to the product;
(10) Exaggerated explanations of the product's scope of application and intended use.
Some label diagrams for reference